IMPORTANT SAFETY INFORMATION
The most commonly reported adverse reactions include vomiting, hair loss, itching, diarrhea, lethargy, dry skin, elevated ALT, and hypersalivation. BRAVECTO PLUS has not been shown to be effective for 2 months in kittens less than 6 months of age. For topical use only. Avoid oral ingestion. The safety of BRAVECTO PLUS has not been established in breeding, pregnant and lactating cats. Fluralaner is a member of the isoxazoline class. This class has been associated with neurologic adverse reactions including tremors, ataxia, and seizures. Neurologic adverse reactions have been reported in cats receiving isoxazoline class drugs, even in cats without a history of neurologic disorders. Use with caution in cats with a history of neurologic disorders. Use with caution in cats that are heartworm positive. The effectiveness of BRAVECTO PLUS to prevent heartworm disease after bathing or water immersion has not been evaluated.
References:
1. BRAVECTO PLUS FOI, NADA 141-518. (Study S15318-00).
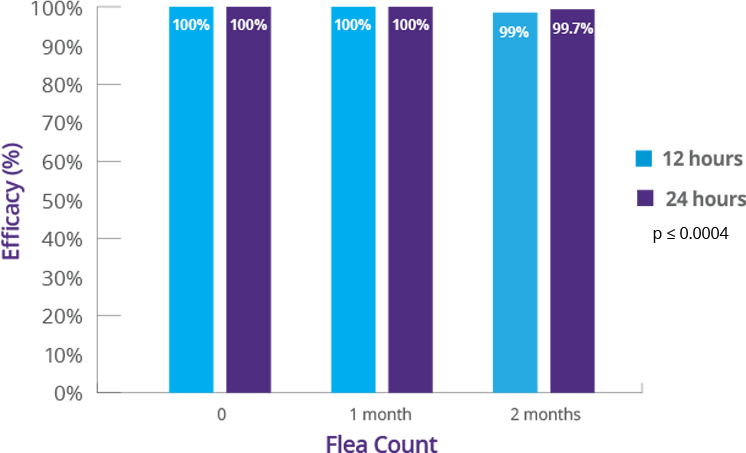
2. BRAVECTO PLUS FOI, NADA 141-518. (Studies S15312-03, 15311-01).
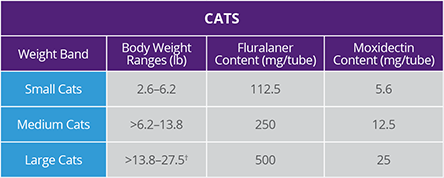
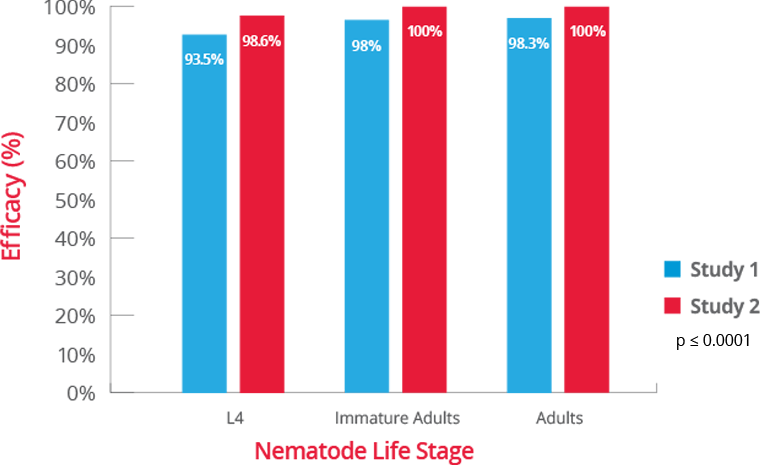
3. BRAVECTO PLUS FOI, NADA 141-518. (Studies S15102-01, S15102-03).
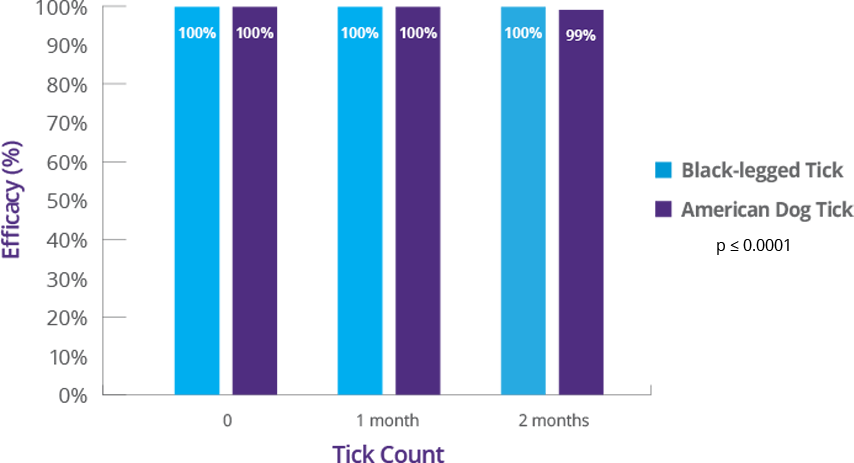
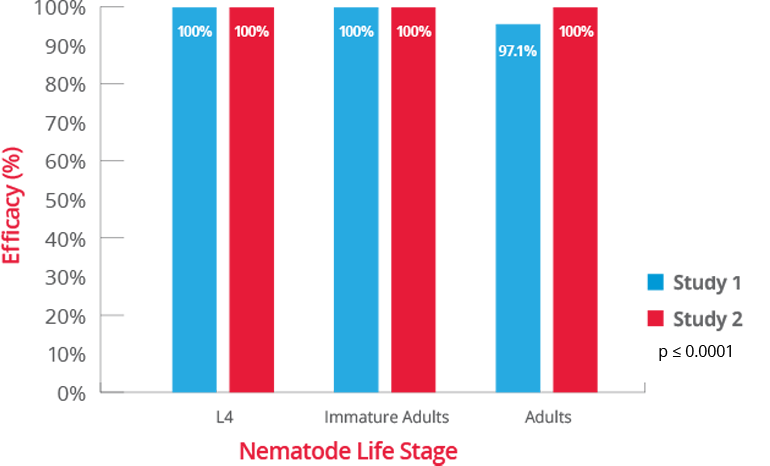
4. BRAVECTO PLUS FOI, NADA 141-518. (Studies S15214-02, S15313-00, S15356-00, S15187-00).
5. BRAVECTO PLUS [prescribing information]. Madison, NJ: Intervet Inc. (d/b/a Merck Animal Health); 2019.
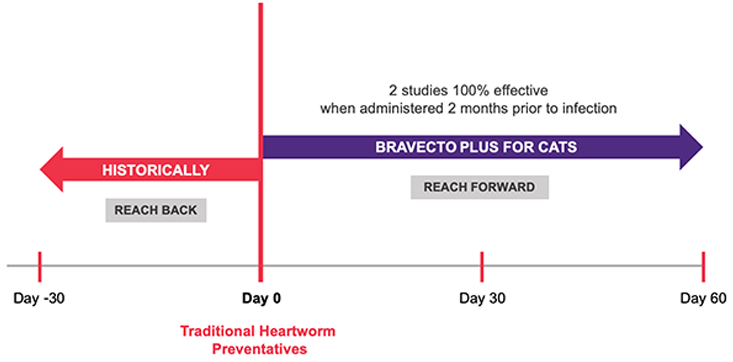
6. BRAVECTO PLUS FOI, NADA 141-518. (Studies S15138-00, S15214-02, S15313-00, S15355-00).
7. Lavan RP et al. Parasites & Vectors. 2017; 10:284.
8. Lavan RP et al. Parasites & Vectors. 2018; 11:581.
9. Brakke Consulting. The US Flea Control and Heartworm Markets. 2018:6-7.
10. CAPC website. Intestinal Parasite Guidelines. Found at www.capcvet.org. Accessed July 11, 2019.
Revolution PLUS is a registered trademark of Zoetis Services LLC.